Procalcitonin as a Predictive Factor for the Clinical Outcome of Patients with Coronavirus Disease 2019
Article information
Abstract
Purpose
The coronavirus disease 2019 (COVID-19) pandemic continues. It has been reported that patients with bacterial coinfection have a higher mortality rate than patients without coinfection. However, there are no clear standard guidelines for the use of antibacterial drugs. Therefore, the purpose of this study was to determine the usefulness of procalcitonin, a specific indicator of bacterial infection, as a biomarker for predicting death in COVID-19 patients.
Methods
This was a retrospective study of confirmed COVID-19 patients (N = 283) between December 2020 and February 2021 who survived or died. Logistic regression analysis was performed to determine whether there was an association between the level of procalcitonin and death. In addition, receiver operating characteristic curve analysis was performed to determine the usefulness of procalcitonin as a predictor of death.
Results
In the non-survivor group, age, the number of patients transferred from a health care center, segment neutrophil ratio, C reactive protein, ferritin, and procalcitonin were significantly higher in the survivor group. In multivariate analysis, procalcitonin was identified as an independent factor associated with death (hazard ratio 6.162, confidential interval 2.285–26.322, p = 0.014). In addition, the predictive power of procalcitonin level and mortality was statistically significant using receiver operating characteristic curve analysis which gave an area under the curve value of 0.823, a cut-off value of 0.05, a sensitivity of 72.2%, a specificity of 87.5% (p < 0.001).
Conclusion
Measurement of procalcitonin and other biomarkers may be useful to determine whether to use or discontinue use of antibacterial drugs in patients with COVID-19.
Introduction
Coronavirus disease 2019 (COVID-19) originated in Wuhan, China and the outbreak was first reported in December 2019. Since then, COVID-19 has continued to spread worldwide. In March 2020 the World Health Organization made an official announcement characterizing COVID-19 as a global pandemic [1]. Vaccination against COVID-19 was started in Korea in March 2021, however, the number of infected people has not decreased. This is due to mutant viruses, breakthrough infections [2–4], and the fact that vaccination against COVID-19 does not prevent infection but does reduce hospitalization rates and deaths due to COVID-19. The overall mortality rate for COVID-19 patients is low (2%), but of those patients, the mortality rate of patients with severe infections, and those who are critically ill is very high (39–72%) [5–9].
Clinically, infection related biomarkers include levels of white blood cells (WBC), segmented neutrophil ratio, procalcitonin (PCT), C-reactive protein (CRP), tumor necrosis factor alpha, and interleukin (IL)-6 levels [10,11]. Among them, PCT is not detectable at less than 0.01 ng/mL in healthy people but increases within 4–12 hours of a bacterial infection. PCT is a specific biomarker of bacterial infection, stimulated by IL-1, IL-6, and tumor necrosis factor alpha, and its production is inhibited by interferon-γ, which increases during viral infection. Several studies of bacterial infections have suggested the use of antibacterial agents according to PCT levels [12–14]. It has been reported that the mortality rate of COVID-19 patients with a bacterial coinfection increases by 1.52–5.82 times. However, treatment recommendations for COVID-19 are regularly being announced, but there is no clear standard for the use of antibacterial agents [5–8,15–18]. Therefore, the purpose of this study was to determine the usefulness of PCT as a biomarker for predicting death in COVID-19 patients.
Materials and Methods
1. Patient selection and data collection
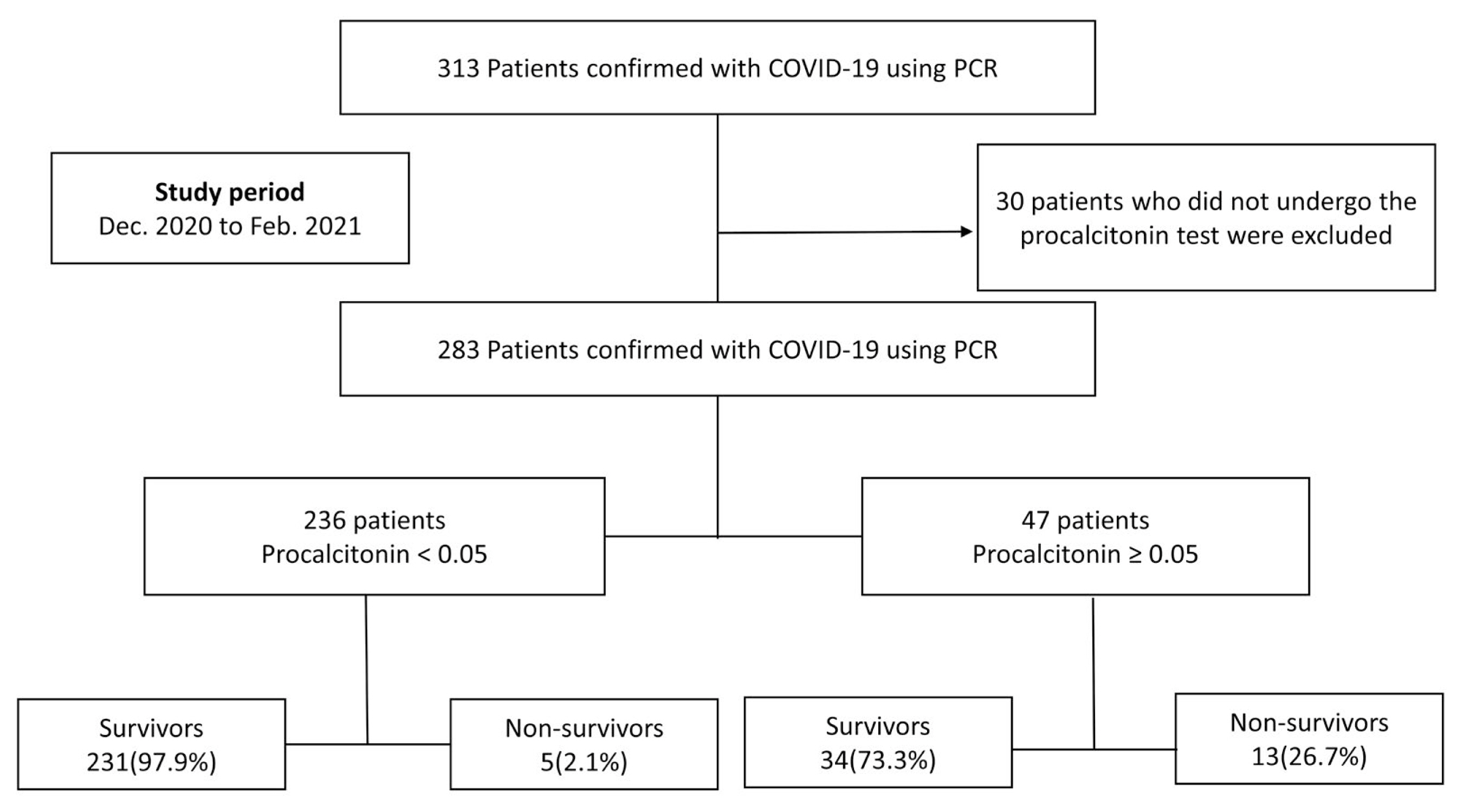
This retrospective study was performed using data from 313 patients with confirmed COVID-19 between December 2020 and February 2021, who were admitted to the National Health Insurance Service Ilsan Hospital (a hospital dedicated to domestic COVID-19). The electronic medical records of the patients were analyzed. After excluding 30 patients who did not undergo the PCT test (Figure 1), 283 patients were included in the study. For the COVID-19 confirmation test, both lateral flow (nasopharyngeal and oropharyngeal smears) and real-time reverse transcription polymerase chain reaction (RdRp, E, ORF1ab, and N) were performed.
2. Hospital settings
The National Health Insurance Service Ilsan Hospital was designated as a COVID-19 regional hospital in December 2021. It has a total of 155 isolated beds under negative pressure ventilation, and 12 intensive care units (ICU). Patients diagnosed with COVID-19 are admitted to a community treatment center if they have no symptoms and are transferred to a regional hospital if their condition worsens. At our institution, the infectious disease department and respiratory internal medicine specialists perform in-hospital severity classification based on patient information before admission. Patients requiring treatment with a high-flow nasal cannula or ventilator care are placed in the ICU. Patients who have a mild oxygen demand and those whose course is expected to deteriorate, go to the moderate symptoms ward, and patients with 2 or more risk factors go to the mild-to-moderate symptoms ward. Patients with 1 or no risk factors are admitted to a mild symptoms ward. In addition, if there is a change in severity of symptoms during hospitalization, the decision of whether to transfer a patient to a different ward is made through the in-hospital communication system.
3. Outcome measures
The primary endpoint of this study was to determine the usefulness of PCT levels in predicting death in COVID-19 patients. The secondary endpoint was the identification of other relevant factors associated with mortality in COVID-19 patients.
4. Statistical analysis
Continuous variables were expressed as mean values and standard errors, and compared using the Student t test. For the analysis of nominal variables, the chi-square test and Fisher’s exact test were used. Logistic regression analysis was used to determine whether the level of PCT was an independent risk factor for death. Furthermore, to confirm the predictive power of PCT, receiver operating characteristic (ROC) curve analysis was performed, and the cutoff value with the highest sensitivity and specificity was confirmed. In order to confirm the ROC curve, PCT < 0.05 ng/mL was defined as an undetectable value. Statistical significance at a 95% confidence interval was defined as a p <0.05. All analyses were performed using SPSS Version 23.0 (SPSS Inc., Chicago, IL, USA).
Results
1. Patient characteristics
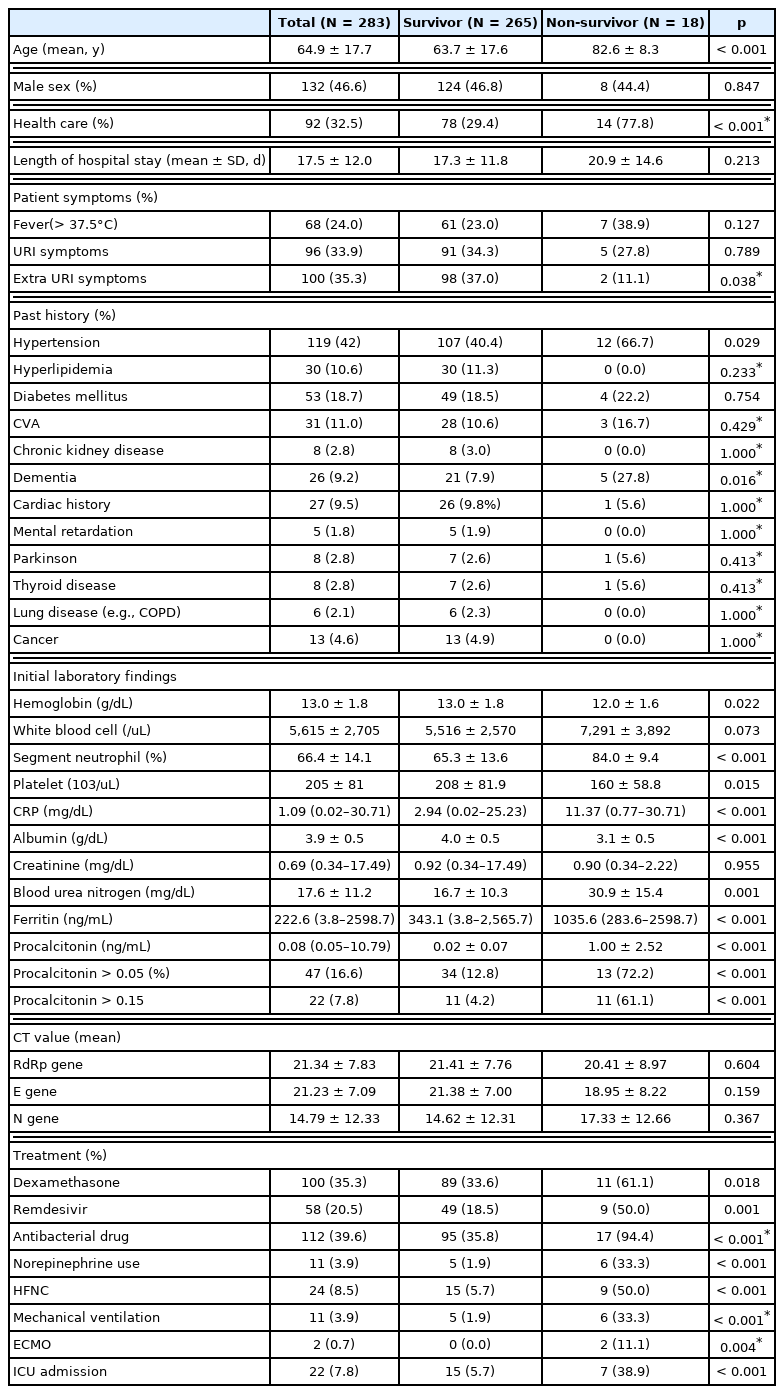
The mean age of the 283 COVID-19 patients analyzed from November 2020 to February 2021 was 64.9 ± 17.7 years, and 132 (46.6%) were male. There were 92 patients (32.5%) who were transferred from other hospitals or healthcare centers. The mean length of their hospital stay was 17.5 ± 12.0 days. There were 18 patients (6.4%) who died. At the time of admission, 24% of patients complained of a fever (≥ 37.5°C); 33.9% had symptoms of upper respiratory tract infection; and 35.3% had symptoms other than upper respiratory tract infections, such as muscle pain, fatigue, and headache. Their medical history included hypertension (42%), diabetes (18.7%), and dyslipidemia (10.6%). At the time of admission, the mean or median (range) laboratory values were as follows: hemoglobin 13.0 ± 1.8 g/dL, WBC 5,615 ± 2,705/μL, segmented neutrophil ratio 66.4% ± 14.1%, platelets 205 ± 81 × 103/μL, albumin 3.9 ± 0.5 g/dL, CRP 1.09 (0.02–30.71) mg/dL, ferritin 222.6 (3.8–2,598.7) ng/mL, and PCT 0.08 (0.00–10.79) ng/mL. Dexamethasone was administered to 100 patients (35.3%), remdesivir to 58 patients (20.5%), and antibacterial agents to 112 patients (39.6%). A high-flow nasal cannula was used in 24 patients (8.5%), mechanical ventilation for 11 patients (3.9%), and extracorporeal membrane oxygenation (ECMO) for 2 patients (0.7%). There were 22 patients (7.8%) who were admitted to the ICU (Table 1).
2. Comparative analysis of survivors and non-survivors
Regarding the characteristics of survivors versus non-survivors, the average age of the non-survivors was significantly higher (63.7 ± 17.6 vs. 82.6 ± 8.3 years, p < 0.001), and the number of non-survivors transferred from healthcare centers was significantly higher (29.4% vs. 77.8%, p < 0.001). The percentage of patients with hypertension (40.4% vs. 66.7%; p = 0.029) and dementia (7.9% vs. 27.8%; p = 0.016) were significantly higher in the non-survivors. The survivor group had more non-respiratory symptoms (37% vs. 11.1%; p = 0.038). Factors significantly different between the 2 groups (survivor vs. non-survivor) in the initial laboratory tests were hemoglobin (13.0 ± 1.8 vs. 12.0 ± 1.6 g/dL; p = 0.022), segmental neutrophil ratio (65.3% ± 13.6% vs. 84.0% ± 9.4%; p < 0.001), platelets (208 ± 81.9 vs. 160 ± 58.8 × 103/μL; p = 0.015), CRP (2.94 vs. 11.37 mg/dL; p < 0.001), albumin (4.0 ± 0.5 vs. 3.1 ± 0.5 g/dL; p < 0.001), blood urea nitrogen (BUN); 16.7 ± 10.3 vs. 30.9 ± 15.4 mg/dL; p = 0.001), ferritin (343.1 vs. 1,035.6 ng/mL; p < 0.001), and PCT (0.02 vs. 1.00 ng/mL; p < 0.001). Dexamethasone (33.6% vs. 61.1%; p = 0.018), remdesivir (18.5% vs. 50.0%; p < 0.001), and antibacterial agents (35.8% vs. 94.4%; p < 0.001) were administered more frequently to non-survivors. The rate of admission to the ICU was higher in the non-survivor group (5.7% vs. 38.9%; p < 0.001). A high-flow nasal cannula (p < 0.001), mechanical ventilation (p < 0.001), and ECMO (p = 0.004) were more commonly used in non-survivors (Table 1).
3. Logistic regression analysis for mortality
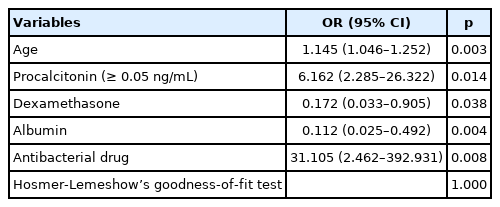
To determine the effect of an elevated PCT level (≥ 0.05 ng/mL) on mortality, a logistic regression model including age, PCT (≥ 0.05 ng/mL), use of remdesivir, use of dexamethasone, transfer from a healthcare center, hypertension, albumin levels, and use of antibacterial agents was constructed. It was confirmed that the model was valid by using the Hosmer-Lemeshow’s goodness-of-fit test (p = 1.000). Age [odds ratio (OR) = 1.145; 95% confidence interval (CI) = 1.046–1.252; p = 0.003], use of dexamethasone (OR = 0.172; 95% CI = 0.033–0.905; p = 0.038), albumin levels (OR = 0.112; 95% CI = 0.025–0.492; p = 0.004), and use of antimicrobials (OR = 31.105; 95% CI = 2.462–392.931; p = 0.008) were determined as independent risk factors related to death (Table 2). An increase in PCT level was identified as a significant risk factor for mortality (OR = 6.162; 95% CI = 2.285–26.322; p = 0.014). An increase in PCT level was also determined to be an independent risk factor associated with death (Table 2).
4. Performance of levels of PCT and other laboratory markers
When ROC curve analysis was performed to confirm the predictive power of PCT, the area under the curve (AUC) was 0.823 (p < 0.001) and the cutoff value was 0.05 ng/mL (Figure 2). Sensitivity and specificity were 72.2% and 87.5%, respectively. The results of the ROC curve analysis for other biomarkers were as follows: CRP (AUC = 0.819; p < 0.001), segmental neutrophil ratio (AUC = 0.872; p < 0.001), ferritin (AUC = 0.873; p < 0.001), age (AUC = 0.837; p < 0.001), WBC (AUC = 0.630; p = 0.065), and BUN (AUC = 0.840; p < 0.001; Figure 3).

Receiver operating characteristic curves for procalcitonin levels between survivors and non-survivors. Procalcitonin area under the curve (AUC) = 0.823; cut-off value = 0.05; sensitivity = 72.2%, specificity = 87.5%, p < 0.001.
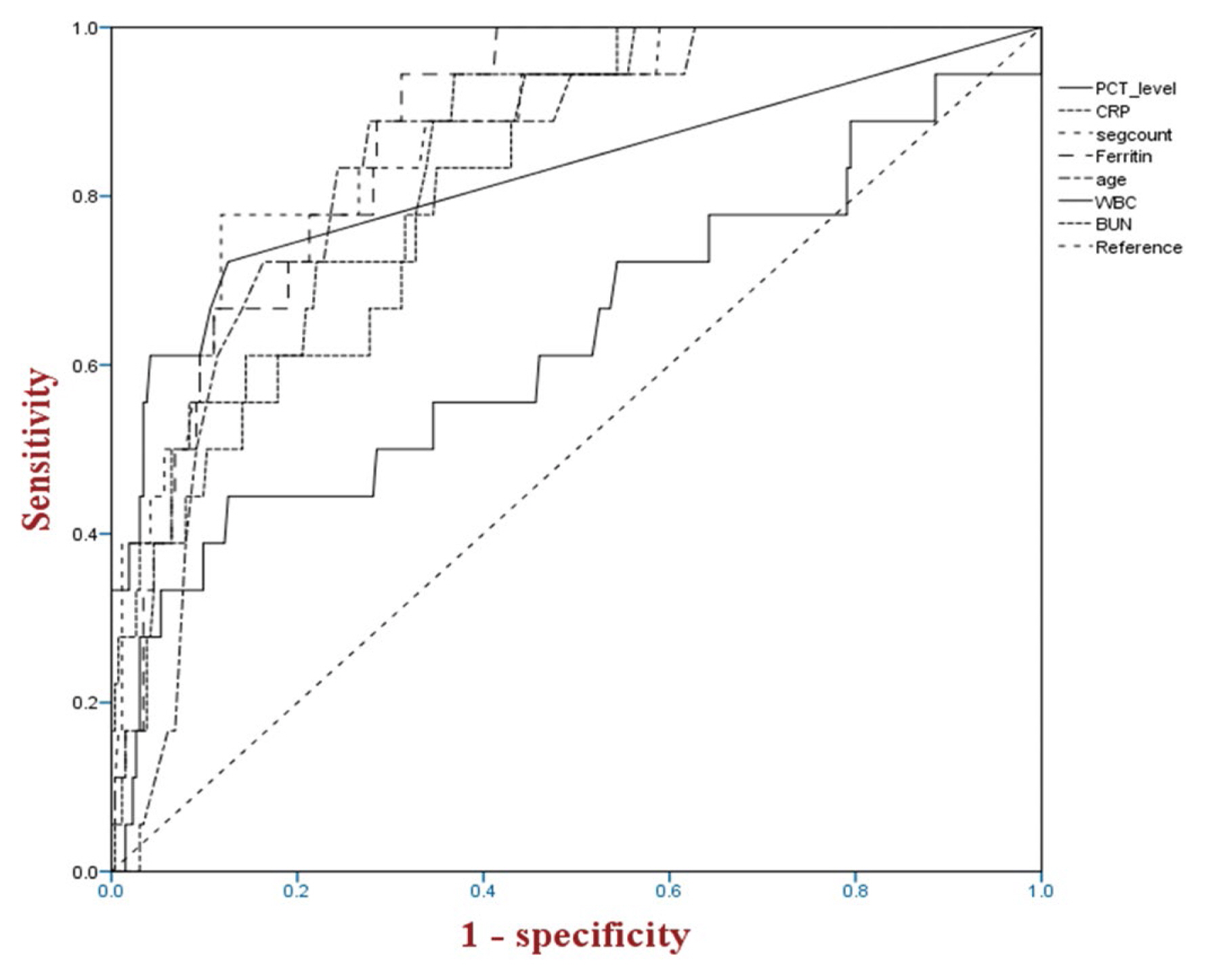
Receiver operating characteristics curves for other biomarkers between the survivors and non-survivors. CRP (ACU = 0.819, p < 0.001); Segment neutropil (AUC = 0.872, p < 0.001); Ferritin (AUC = 0.873, p < 0.001); Age (AUC = 0.837, p < 0.001); white blood cell (AUC = 0.630, p = 0.065); BUN (AUC = 0.840, p < 0.001).
5. Comparison according to PCT level
A total of 236 patients (83.4%) had a PCT level < 0.05 ng/mL, and 47 patients (16.6%) had an elevated PCT level (≥ 0.05 ng/mL). Patients with elevated PCT levels were significantly older (p < 0.001), and the rates of admission to the ICU (4.2% vs. 25.5%; p < 0.001) and transfer from a healthcare center (27.5 vs. 57.4%; p < 0.001) were also significantly higher. Laboratory findings showed that the segmental neutrophil ratio (p < 0.001), CRP level (p < 0.001), creatinine level (p = 0.019), BUN (p < 0.001), and ferritin level (p < 0.001) were significantly higher in the elevated PCT group, and the platelet (p = 0.002) and albumin level (p < 0.001) were significantly lower. A higher proportion of patients with elevated PCT had a medical history of hypertension (39.4% vs. 55.3%; p = 0.044) and chronic renal failure rate (0.8% vs. 12.8%; p < 0.001) (Table 3). Treatment with dexamethasone (29.2% vs. 66.0%; p < 0.001), remdesivir (14.8% vs. 48.9%; p < 0.001), and antibacterial agents (33.5% vs. 70.2%; p < 0.001) was significantly higher in the elevated PCT group. High-flow nasal cannula (3.8% vs. 31.9%; p < 0.001), mechanical ventilation (1.7% vs. 14.9%; p < 0.001), and ECMO (0% vs. 4.3%; p = 0.027) were also commonly used in the elevated PCT group (Table 3). The mortality rate (2.1% vs. 27.7%; p = 0.001) and length of hospital stay (16.5 ± 11.5 vs. 23.1 ± 13.9 days; p = 0.003) were also significantly higher in the elevated PCT group (Table 3).
Discussion
Multivariate analysis of mortality-related factors were performed to determine the usefulness of the level of PCT in patients with COVID-19 for predicting death. An increase in PCT level resulted in a high OR (6.599). These results were similar to those of a previous study of 1,527 COVID-19 patients (hazard ratio, 4.933] [19]. In addition, a previous study reported that PCT levels were low (≤ 1.00 ng/mL) in patients with viral pneumonia, including severe acute respiratory syndrome coronavirus-1 (SARS-CoV-1) infection, but increased in patients with bacterial and fungal pneumonia [19–23]. Considering that COVID-19 has a clinical picture similar to SARS-CoV-1, it may be inferred that coinfection with COVID-19 and bacteria may have occurred in patients with elevated PCT levels in this study. Several studies on COVID-19 have reported that the use of antimicrobials in patients without bacterial coinfection is associated with a worsening of the patient’s clinical outcome [19,24]. Recently, antiviral agents such as baricitinib and nirmatrelvir have been used, however, in the actual treatment of COVID-19 patients, dexamethasone, remdesivir, and monoclonal antibodies constitute the main axis, though most patients depend on symptomatic therapy. Therefore, determining the most appropriate use of antibacterial agents is important. Most studies and guidelines on the treatment of COVID-19 recommend considering the use of antimicrobials depending on the severity of the disease [17,18,25–28]. In our institution, as per the guidelines, most clinicians prescribe antibacterial drugs according to the severity of the disease, and 34% of patients who did not have elevated PCT levels were administered antibacterial drugs.
In this study, the use of antibacterial drugs was identified as a factor that significantly increased the risk of death in multivariate analysis. Based on the increase in PCT levels and the use of antibacterial drugs, patients were classified into 4 groups and comparatively analyzed. The mortality rate was higher when the PCT level was < 0.05 ng/mL, and antibacterial drugs were used than when PCT was < 0.05 ng/mL and no antibacterial drugs were used. In fact, the severity of disease was very high in the group that received antibacterial drugs (Table 4). Improper selection and long-term use of antibacterial drugs are major causes of multidrug-resistant organisms, with 30–50% of antibacterial drug resistance cases due to the inappropriate use of antibacterial drugs, such as errors in drug selection or duration of use [29]. Multi-resistant bacterial infection is an important factor for poor prognosis and limits the choice of antibacterial agents [30]. Previous studies have reported that approximately 50% of COVID-19 patients who died had bacterial coinfections [22,31]. Antibacterial drugs are being used only for severely ill patients, following the recommendations for the use of antimicrobials in COVID-19 patients. If antibacterial drugs are used according to the severity of COVID-19, there is a possibility that antibacterial treatment may be delayed and the condition may worsen in patients with mild or moderate bacterial coinfection.
In conclusion, the early detection and treatment of bacterial coinfections in COVID-19 is important. Therefore, antibacterial agents in COVID-19 patients should be used cautiously. Furthermore, checking the possibility of bacterial coinfection by measuring PCT before administration may be considered clinically effective. In this study and previous studies, the use of antibacterial drugs as a risk factor for poor prognosis is thought to be due to delays in therapy as well as selection bias in the present retrospective study. In previous studies and meta-analysis, PCT was analyzed as a predictive factor of mortality and severity [21,32–36]. In addition, in 1 study, PCT was analyzed as an important predictive factor of bacterial coinfection according to culture results [36]. However, in the previous studies, there was no case of analyzing the correlation between the use of antibacterial agents according to the PCT value. Even this current study is a single-center study, but this includes all patients from mild to severe. In addition, the independent effect of use of antibacterial agents on mortality was analyzed (Table 2), and the use of anti-bacterial agent according to the PCT value was also analyzed (Table 4). In 27.5% of patients, antibacterial agent was used despite a PCT level of < 0.05 ng/mL, and in 4.9% of patients, antibacterial agent was not used even though the PCT level was ≥ 0.05 ng/mL. Among patients with a PCT level of < 0.05 ng/mL, when compared according to whether antibacterial agents were used, there were significant differences in clinical outcomes such as ICU admission, length of hospital stay, and mortality between the 2 groups (supplement table). In addition, direct physical examination is limited because the treatment environment for COVID-19 follows the principle of minimum personnel and contact for the safety of medical staff. Considering these points, it is expected that it will be useful to some extent to measure the level of PCT and other biomarkers to determine whether to use or discontinue use of appropriate antibacterial drugs in patients with COVID-19.
This study had several limitations. Firstly, this was a single-institution, retrospective study. Antibacterial drugs were administered at the discretion of the clinician. Therefore, the criteria for moderate severity of COVID-19 symptoms were unclear. Secondly, a small number of COVID-19 patients were included in the study, and because the mortality rate was not high, the number of deaths in the patient group was small (18), which means that the statistical power was weak. Despite these limitations, this study is meaningful in that it demonstrates the usefulness of the level of PCT in predicting bacterial coinfection in COVID-19 patients and determining whether antibacterial drugs should be used. A larger prospective study is needed to confirm these findings in the future.
Supplementary Material
Supplementary material is available at https://www.jacs.or.kr.(doi; https://doi.org/10.17479/jacs.2022.12.2.53)
Acknowledgments
This study received research funding from the National Health insurance Service Ilsan Hospital.
Notes
Author Contributions
Conceptualization: JYJ. Methodology: JYJ and SHL. Formal investigation: JYJ and SHL, TYC, JML, KYL and, KHP. Data analysis: SHL, JYJ, TYC, and JML. Writing original draft: SHL, Writing - review and editing: SHL, JYJ, TYC, JML, KYL, and KHP
Conflicts of Interest
Ji Young Jang has been the associate editor of Journal of Acute Care Surgery since March 2017, but had no role in the decision to publish this Original Article. No other potential conflict of interest relevant to this article was reported.
Ethical Statement
This study was approved by the institutional review board of the National Health Insurance Service Ilsan Hospital and complied with the Declaration of Helsinki (NHIMC no.: 2021-03-009). This study was conducted retrospectively, and informed consent was waived. All personally identifiable information was collected anonymously.
Data Availability
All relevant data are included in this manuscript.