Tranexamic Acid in Trauma Management: A Review of Evidence
Article information
Abstract
Hemorrhage is the leading cause of death in trauma patients and trauma induced coagulopathy (TIC) is a major contributor to bleeding mortality. TIC has a diverse pathophysiology triggered by injury and hypoperfusion, including platelet dysfunction, endotheliopathy, fibrinogen or thrombin abnormalities, and dysregulated fibrinolysis. Early fluid resuscitation, appropriate blood transfusion, and definitive control of bleeding are essential components of initial management for TIC. Additionally, tranexamic acid (TXA), an antifibrinolytic agent, has emerged as a potential adjunctive therapy following the 2010 landmark trial that demonstrated the benefit of early administration of TXA in reducing trauma patient mortality (CRASH-2). This review provides an analysis of the current literature on the use of TXA in trauma patients. It critically evaluates the evidence on the effect of TXA on TIC and other clinical outcomes, emphasizing the time-sensitive nature of TXA administration and the variation of its effect depending on the severity and location of injury. It also discusses the optimal dosage, timing, and safety of TXA, as well as the challenges and limitations of existing studies. Furthermore, it highlights the importance of individualized treatment approaches based on the fibrinolysis status of TIC and the value of goal-directed therapy guided by viscoelastic hemostatic assays for the appropriate use of TXA.
Introduction
Hemorrhage is the leading cause of early mortality in cases of major trauma [1], with coagulation abnormality manifesting in approximately 25% of patients with severe trauma, and higher mortality in patients with the coagulopathy [2].
The cornerstone of initial management includes early fluid resuscitation, prompt and appropriate blood transfusion, and definitive control of bleeding. Following the “Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage” (CRASH)-2 study and the CRASH-3 study, the administration of tranexamic acid (TXA) has emerged as a potential adjunctive strategy to standard resuscitation measures for trauma induced coagulopathy (TIC) and hemorrhagic shock [3,4]. However, despite its widespread application, the current understanding of TXA still remains limited, particularly regarding its therapeutic efficacy, indications for use, safety dosage, organ-specific effects (such as renal) and potential side effects. While its effectiveness in various scenarios such as pre-surgery or nontraumatic bleeding has been explored, many aspects remain to be elucidated. In addition, many recent studies have shown the existence of a fibrinolysis phenotype, which provides an important theoretical background for the more accurate, effective, and safe use of TXA as an antifibrinolytic agent [5]. This review aimed to shed light on the evidence supporting TXA use across diverse clinical conditions and environments, including trauma. Particularly, the review examined the judicious use of TXA in relation to the clinical phenotype of coagulopathy as revealed through recent studies with viscoelastic assays.
Discussion
1. TIC
1.1. Historical research
Acute trauma coagulopathy, now called TIC, has emerged as a concern. It was reported in 2003 that significant coagulopathy occurred in 25% of severely injured patients, regardless of volume resuscitation [2]. This acute coagulopathy of trauma was associated with systemic hypoperfusion, that is hemorrhagic shock, and was characterized by anticoagulation and hyperfibrinolysis [6].
Out of a Trans-Agency Coagulopathy in Trauma Workshop in April 2010 this meeting presented a consensus that the term “trauma induced coagulopathy” would be employed to describe commonly what was previously referred to as acute traumatic coagulopathy [7].
1.2. Epidemiology
In several studies, the incidence of TIC has been reported to be about 24–40% in severe trauma patients [8–11]. In general, children develop TIC later and less frequently than adults, and older people are more vulnerable to TIC [12,13].
1.3. Pathophysiology
The underlying pathophysiological mechanisms are complex and remain poorly elucidated. However, it is known that acute coagulopathy in severe trauma patients is caused by the combined effects of tissue damage and hypoperfusion, that is, injury and hemorrhagic shock [14]. Subsequent to the initial injury and hypoperfusion, a cascade of biochemical and humoral responses are triggered, both locally at the site of injury and systemically throughout the body. Pathophysiological bases of these responses can be summarized as endothelial cell dysfunction and platelet dysfunction, i.e., dysfunction of cell-based hemostasis, fibrinogen depletion, and dysregulated fibrinolysis [7,15,16]. Within this mechanism, primary fibrinolysis dysfunction provides a physiological plausibility for the administration of hyperfibrinolytic agents in the initial treatment of TIC in patients with hemorrhagic shock, just as the CRASH-2 trial provided clinical relevance [17].
2. Mechanism of action and pharmacokinetics of TXA
TXA (trans-4-aminomethyl cyclohexane carboxylic acid) is a synthetic lysine analogue made by Japanese physiologist, Utako Okamoto in 1962 [18]. TXA exhibits antifibrinolytic function by interfering with the action of the plasminogen activator by reversibly binding to the lysine binding site of plasminogen thereby blocking interactions with fibrin and subsequent clot breakdown and stabilizing a previously formed clot [19]. In addition, TXA also exhibits secondary effects by inhibiting plasmin. TXA improves platelet function and inhibits plasmin-induced platelet activation which facilitates clot stabilization [20].
Plasmin is also a known activator of inflammatory cells, cytokines, and immune mediators and produces proinflammatory effects by binding to and activating monocytes, neutrophils, platelets, and endothelial cells, and releasing lipid mediators and cytokines [21]. So, TXA may attenuate the intense inflammatory response by inhibiting plasminogen or plasmin-mediated inflammation [22]. This anti-inflammatory effect of TXA may also result in a reduction in multiple organ failure (MOF) in severely injured patients [23].
The pharmacokinetics of TXA in a healthy adult reveals its peak concentration by 60 minutes following intravenous (IV) administration and has a half-life of approximately 2 hours. An antifibrinolytic dosage remains in serum and tissue for up to 8 and 17 hours, respectively [24].
Furthermore, it is crucial to consider the alterations in pharmacokinetics depending on the administration route of TXA and its subsequent impact on efficacy. Intravenous administration of TXA is predominantly employed in clinical settings for severe trauma patients presenting with shock and coagulopathy. However, intramuscular (IM) administration is also considered, particularly during prehospital stages or in an Emergency Room where IV access may not be readily available. This method provides a rapid delivery solution in urgent situations. Several animal studies and trials involving normal adult participants have reported no significant difference between IM and IV administration in terms of dosage and time required to achieve effective blood concentrations [25,26]. Therefore, based on these findings, it can be inferred that IM administration is a viable alternative under certain circumstances [27]. However, the pharmacokinetics of this drug in trauma patients may be different to that observed in normal adults, and the clinically appropriate therapeutic dose for patients with severe trauma or hemorrhagic shock has not yet been clearly established.
3. Timing of administration of TXA
While the optimal TXA administration (timing and duration) in trauma patients remains undetermined, the CRASH-2 study significantly contributed to our understanding of this issue. This landmark randomized placebo-controlled trial of the effect of TXA on mortality in 20,211 trauma patients showed TXA safely reduced the risk of death in trauma patients who were bleeding, without definitive risk of thromboembolic adverse effects [3]. This survival benefit is only evident in patients in who were treated within 3 hours of their injury [heart rate (HR) ≤ 3 hours = 0.78, 0.68–0.90; HR > 3 hours = 1.02, 0.76–1.36]. Initiation of TXA treatment within 3 hours of injury reduced the hazard of death due to bleeding on the day of the injury by 28% (HR = 0.72, 0.60–0.86) [28]. The results suggest that TXA should be given as soon as possible after injury to trauma patients who are bleeding, and that its use should be avoided beyond 3 hours post injury. A study by Ggayet-Ageron et al [29] reinforced the significance of early TXA administration as their results indicated that survival benefits decreased by 10% for every 15-minute delay beyond the initial 0–3-hour window. This led many treament guidelines based on an evidence-based consensus approach which included the recommendation that TXA be administered as soon as possible and within 3 hours of injury [30].
The positive outcomes associated with timely TXA administration extend beyond trauma cases. Similar benefits have been observed in the context of postpartum hemorrhage [31], suggesting that the importance of administration timing may be a general principle in managing critical bleeding scenarios.
The evidence, primarily drawn from the CRASH-2 trial and subsequent research, consistently supports the early initiation of TXA treatment, ideally within 1 hour of injury. These findings have profound implications for the clinical management of trauma patients, underlining the life-saving potential of timely TXA intervention and inevitably led to interest and research into the prehospital use of this drug.
4. TXA: prehospital use
The results of research on the early administration and effectiveness of TXA were ultimately related to the interest in the effect of the prehospital use for trauma patients. Stein et al [32] reported the results of the prospective, multicenter, observational study of the assessment for the benefit effect of on-scene IV administration of TXA in 2018. In a prospective, multicenter observational study comparing trauma patients who received prehospital TXA with a control group (without TXA), it was observed that early TXA administration led to clot stabilization, reduced fibrinolytic activity, and a significant decrease in production of fibrin degradation products (D-dimer). Specifically, viscoelastic tests showed that maximum clot firmness (MCF) did not change from on-scene to the Emergency Department in the TXA group, while MCF reduced in the control group. This study indicates that prehospital TXA administration plays a crucial role in enhancing coagulation and minimizing fibrinolysis in trauma patients [32].
A multicenter randomized clinical trial on the effects of prehospital use of TXA on approximately 900 injured patients in 2021 [“Study of Tranexamic Acid During Air Medical Prehospital Transport” (STAAMP) trial] showed significantly lower 30-day mortality in the subgroup of the patients administered within 1 hour after injury (4.6% vs 7.6%; p < 0.002) and with severe shock (18.5% vs 35.5%; p < 0.003) [33].
However, in a study on prehospital TXA administration in 1,827 traumatic brain injury (TBI) patients conducted in the Netherlands, 30-day mortality was poor in the TXA administered group for severe isolated TBI patients (OR, 4.49; 95% CI, 1.57–12.87; p = 0.005) [34] and Rowell et al [35] also reported results that did not support effects of prehospital TXA administration on patients with severe traumatic brain injury.
In a recent randomized controlled trial (RCT), “Pre-hospital Anti-fibrinolytics for Traumatic Coagulopathy and Haemorrhage-Trauma” (PATCH trial) on the effect of the pre-hospital use of TXA on survival benefit among 1,310 adults with major trauma and suspected trauma-induced coagulopathy, prehospital administration of TXA followed by infusion over 8 hours did not result in better patient survival or favorable functional outcome at 6 months compared with the placebo [36]. Research on the prehospital use of TXA has produced a variety of results, indicating that a consensus on its effects has not yet been established.
5. TXA: use for non-trauma patients
Considerable research on the effects of TXA on bleeding, mortality, and the occurrence of several types of thromboembolic events has been conducted not only in trauma patients but also in various non injury related clinical situations where bleeding occurs such as post operative bleeding, postpartum hemorrhage, nontraumatic gastrointestinal bleeding, and nontraumatic cerebral hemorrhage. A randomized study in 2017 on the effectiveness of TXA administration in 4,662 cardiac surgery patients did not show mortality benefit (relative risk, 0.92; 95% CI 0.81–1.05; p = 0.22), but a reduction in the amount of transfused blood products, and occurrence of postoperative hemorrhagic complications was observed [37]. A randomized trial on the usefulness of TXA on major bleeding and the occurrence of complications among 9,535 patients undergoing noncardiac surgery showed the incidence of the major bleeding outcome was significantly lower with TXA than the placebo [38]. A small, randomized trial on the preoperative administration of TXA in aortic aneurysm surgery reported that preoperative TXA did not reduce intraoperative blood loss or blood transfusion but may reduce postoperative blood loss without increasing adverse effects. However, it should be taken into account that the dose of TXA used in this study was lower than the dose in other general studies (loading dose of 500 mg and a continuous infusion of 250 mg/h) [39].
For the orthopedic surgery, TXA formulations were superior to the placebo in terms of decreasing blood loss and risk of transfusion after total knee arthroplasty surgery [40].
Two well-designed RCTs analyzed the effects of TXA on postpartum hemorrhage outcomes [31,41]. In a RCT by Ducloy-Bouthors et al [41], the effectiveness of 4 g TXA on postpartum hemorrhage was evaluated. The trial included 144 women with postpartum hemorrhage of more than 800 cc, who were randomly assigned to receive either 4 g TXA or a placebo. The results showed that the TXA group had significantly lower blood loss within the first 6 hours of treatment than the control group (median 173 mL vs 221 mL, p = 0.041) [41]. The WOMAN trial published in 2017, a large-scale RCT, investigated the effect of TXA administration on postpartum hemorrhage [31]. The trial enrolled 20,000 women with postpartum hemorrhage and randomly assigned them to receive either 1 gm of TXA or a placebo. The results showed that TXA significantly reduced the risk of death due to bleeding (1.5% vs 1.9%, p = 0.045) although it did not affect the overall mortality rate [31].
In conclusion, TXA is a promising hemostatic agent that has been shown to reduce bleeding and mortality in various nontraumatic clinical situations such as cardiac, noncardiac, orthopedic, and obstetric surgeries. However, the optimal dose, timing, and duration of TXA administration remains unclear and requires further investigation. Additionally, the safety and efficacy of TXA in nontraumatic cerebral and gastrointestinal hemorrhages, needs to be established by performing large-scale randomized trials, because the benefits in terms of mortality and VTE incidence stability need to be elucidated [42].
6. TXA: use in trauma
The CRASH-2 study, despite its limitations, is often regarded as the pioneering study that sparked this discourse. This trial, which showed a 14.5% reduction in 28-day mortality in the TXA group compared to 16% in the placebo group, has impacted TXA use in trauma patients [3]. After this study, the advantages and disadvantages of TXA use in trauma patients have been thoroughly explored.
While the study conducted by Morrison et al in 2012, was retrospective in nature, it nonetheless offered valuable insights into the efficacy of TXA administration [43]. The findings underscore key considerations in identifying individuals who would most benefit from TXA treatment. In this comparative analysis of 896 combat injury patients with or without the administration of TXA, the researchers reported a significant correlation between the use of TXA and survival rates. The odds ratio was remarkable 7.228 [95% CI (3.016–17.322)]. Furthermore, the incidence of coagulation disorders was notably influenced by TXA usage. This association was stronger in the group of patients who underwent massive transfusion. Therefore, TXA administration should be considered in addition to blood transfusions group as part of a resuscitation strategy following severe injury and hemorrhage.
In the “Military Application of Tranexamic Acid in Trauma Emergency Resuscitation” (MATTERs)-2 study of the effects of TXA and cryoprecipitate on mortality in a larger group of patients than in the MATTERs study, the mortality benefit was greatest in patients who received both TXA and cryoprecipitate [44]. This may mean an additional effect of fibrin on the antifibrinolytic function of TXA, or another synergic effect of TXA and fibrin, but more research is needed.
Cole et al [23] reported a significant association of TXA administration with a reduction in all-cause mortality and MOF in injured patients with shock, and the result of the MATTERs study revealed the most severely injured group benefited the most from the administration of the TXA. These are consistent with the results of subgroup analysis by systolic blood pressure (≤ 75 mmHg, 76–89 mmHg, and > 89 mmHg) in the CRASH-2 study.
However, studies on the benefits of TXA in traumatic brain injury are slightly different. The CRASH-3 trial [4] published in 2019, was a randomized, placebo-controlled trial conducted in 175 hospitals across 29 countries. This trial aimed to assess the effect of TXA treatment within 3 hours of injury in the 12,737 patients with isolated traumatic brain injury who had a GCS of 12 or lower and no major extracranial bleeding. Among them, blind randomization was performed resulting in patients who received TXA [6,406 (50.3%) or the placebo 6,331 (49.7%)], of whom 9,202 (72.2%) patients were treated within 3 hours of injury. This study did not show statistically meaningful differences in the head injury-related death between 2 groups [18.5% in the TXA group versus 19.8% in the placebo group; RR 0.94 (95% CI 0.86–1.02)]. However, this study did show that the effect of TXA varied depending on the degree of brain injury. The risk of head injury-related death reduced with TXA treatment in patients with mild-to-moderate head injury [RR 0.78 (95% CI 0.64–0.95)] but did not in patients with severe head injury [0.99 (95% CI 0.91–1.07)]. In addition, early treatment with TXA was more effective than later treatment in patients with mild to moderate head injury (p = 0.005) but time to treatment had no obvious effect in severe head injury group (p = 0.73). The risk of vascular occlusive events [RR 0.98 (95% CI 0.74–1.28)] and the risk of seizure [1.09 (95% CI 0.90–1.33)] were similar in the TXA and placebo groups. If we recall the results of subgroup analysis in the CRASH-2 study, the results of the CRASH-3 study effect of TXA according to the severity of brain damage were consistent with each other. Furthermore, the finding that early TXA administration was more effective than late administration for survival was also consistent with the results of CRASH-2 [3,4]. In addition to the CRASH-3 study [3], several studies on the effect of TXA in TBI patient groups showed similar results.
In a nested study of more than 200 patients who enrolled in the CRASH-2 study, the authors reaffirmed the results of the CRASH-2 study in the hemorrhagic TBI patient group [45]. Treatment with TXA was associated with a 7% reduction in all-cause mortality, a 5.6% reduction in head injury-related mortality, and a 13.3% reduction in overall poor outcomes. The trial team reported a 7% reduction in all-cause mortality, a 5.6% reduction in head injury-related mortality, and a 13.3% reduction in overall poor outcomes with TXA, and showed a high probability of decreased hemorrhage growth, intracranial mass effect, and new hemorrhage development when compared with matched controls.
Rowell et al [35], in a post hoc study of the CRASH-3 study [4], compared the effects of 1 g and 2 g TXA groups in the pre-hospital setting for patients with mild to moderate TBI. No difference in 28-day mortality and neurological outcome was observed. However, a trend in mortality reduction was observed in the group administered 2 gm of TXA, but seizure risk also increased. This study provides us with important information regarding the appropriate dosage of TXA for TBI patients.
However, not all studies supported the results of the CRASH-2 study in patients with nontraumatic brain hemorrhage, especially aneurysmal subarachnoid hemorrhage, and short-term antifibrinolytic therapy with TXA reduced the risk of rebleeding but did not improve clinical outcome [46].
In conclusion, TXA is an effective hemostatic agent that has been shown to reduce mortality and bleeding in trauma patients, especially when administered early, and in patients with shock or requiring massive transfusion. However, the effect of TXA may vary depending on the severity and location of injury such as traumatic brain injury. Further research is needed to determine the optimal dose, timing, and duration of TXA administration, as well as the safety, and efficacy of TXA in different types of injuries.
7. Dosage of TXA in trauma patients
The TXA dose currently administered to trauma patients or used in research was the empirical dose based on the CRASH-2 trial (1 g/IV for 15 minutes and 1 g/IV over 8 hours). In this trial, the dosage decision was relatively simple and was based on the 2007 Cochrane review paper [47]. However, there is still much debate as to the appropriate TXA treatment dose.
The antifibrinolytic effect of TXA primarily results from the inhibition of hyperfibrinolysis induced by tissue plasminogen activator (tPA). This is achieved through binding to lysine-binding sites on plasmin and plasminogen, thereby preventing the degradation of fibrin molecules. In keeping with the antifibrinolytic function of TXA, a plasma concentration of 10 μg/mL or 100 μg/mL TXA were required to reach 80% and 98% inhibition, respectively [48].
According to another in vitro study, a concentration of 31 μg/ mL TXA was required to fully inhibit tPA-induced fibrinolysis [49].
There was an experimental study on the appropriate concentration of TXA in the blood to achieve antifibrinolytic function in trauma patients with hyperfibrinolysis. TXA was administered at a median time of 43 minutes after trauma and the plasma TXA level measured was 28.7 [21.5–38.5 (8.7–89.0)] μg/mL on arrival at hospital, which was 57 [43–70 (20–148)] minutes after pre-hospital administration of the drug. It was reported that 20% of the trauma patients who received TXA (1 g) at the scene within 1 hour of injury, had suboptimal levels of TXA with concentrations below 20 μg/mL in 20% of the patients [50]. Therefore, the administered dose of TXA in the CRASH-2 trial, 1 g IV bolus followed by additional administration of 1 g over 8 hours, may be judged to be somewhat appropriate.
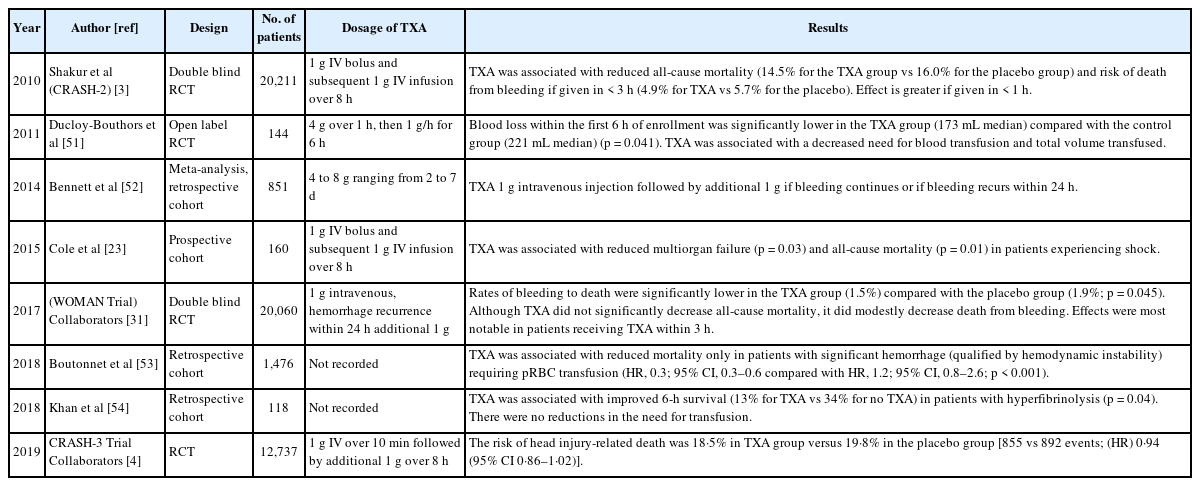
However, apart from these research results, as shown in Table 1 [3,4,23,31,51–54], There are many other studies on effective TXA administration doses according to various situations, and most of these studies seem to be based on empirical decisions or borrowing doses used in existing clinical studies. Dowd et al [55] reported that a dosage of 2 g of TXA administered over 8 hours was effective in the group of non-trauma patients undergoing cardiopulmonary bypass surgery. And another non-trauma, cardiac surgery study indicated an increase in neurotoxicity when the TXA dosage more than twice that in the CRASH-2 trial was administered [56].
While it is plausible to hypothesize that high doses of TXA may induce adverse effects, there is limited research addressing the threshold dose at which these effects occur. Consequently, further studies are needed to determine the optimal dosage of TXA to consider the potential for adverse events, such as seizures, and thrombotic incidents, particularly when administering high doses.
8. Fibrinolysis phenotype in TIC and TXA
In 2014, Moore et al [57] reported that there was a spectrum for fibrinolysis status in groups with TIC [58]. Trauma patients with injury and shock can be divided into 3 phenotypes, including hyperfibrinolysis HF, fibrinolysis shutdown (SD), and physiological fibrinolysis status group, depending on the fibrinolysis status [57,58]. These observations continue to raise the question as to whether TXA should be used selectively following injury based on the degree of shock. The existence of distinct phenotypes based on the fibrinolysis status of injured patients suggests that the effectiveness of TXA, an antifibrinolytic agent, may vary. It is hypothesized that TXA would be particularly beneficial for patients exhibiting HF, while potentially exacerbating adverse effects in the SD group. This hypothesis has been tested and accepted by subsequent studies.
In a study on the effect of TXA administration in trauma patients with injury and shock (3 phenotypes) where each group was confirmed using TEG analysis, it was reported that fibrin clot strength increased in the HF group, but not in the SD or physiological fibrinolysis group, which was predicable based on the mechanism of action of TXA [59].
In addition, Khan et al [54] investigated the effects of TXA on mortality, transfusion volume, hemostasis, rebleeding, and thrombosis complication in 680 severely injured patients and observed that the administration of TXA increased 6-hour survival in patients with HF. In addition, Meizoso et al [60] reported patients who received TXA were at increased risk of fibrinolysis shutdown SD compared with patients who did not receive TXA.
Moreover, fibrinolytic phenotypes can change over time after injury. Robert et al [61] highlighted these temporal changes in the 3 phenotypes within 24 hours following injury and demonstrated that HF patients initially exhibiting a higher frequency of phenotypes may transition to other types such as SD or physiologic fibrinolysis phenotype within 24 hours. Furthermore, the study revealed that the persistence of the SD phenotype for more than 24 hours was associated with increased mortality.
Coats et al [62] investigated the plasma level of tPA and plasminogen activator inhibitor type 1 (PAI-1) in major trauma patients and illustrated an inverse relationship in the temporal changes of median tPA and PAI-1 concentrations. Initially, elevated tPA levels rapidly declined, while initial PAI-1 levels exhibited a gradual increase. The results of this study suggest the presence of a natural antifibrinolytic system that lags by several hours behind the natural profibrinolytics. These findings support early administration of TXA, to address the poor outcomes associated with delayed administration of TXA use as observed in the previous study [3].
9. Adverse effects of TXA
When considering the use of TXA for the bleeding patients, the risk of adverse events must also be considered. TXA is usually well tolerated and generally considered safe at the empirical dosage. However, TXA can provoke several adverse effects including nausea, diarrhea, drug eruption, renal injury, seizure, and several thromboembolic events like deep vein thrombosis, cerebrovascular infarction, myocardial infarction, and pulmonary embolism. Most of these are mild or subtle, but some can be fatal. In the context of these potentially fatal side effects, there is a scarcity of research findings concerning the dosage, route of administration, and circumstances related to the risk of occurrence. This highlights the need for further investigation to ensure safe and effective use of TXA. The incidence of thrombotic events among bleeding patients who receive TXA is not fully known even though many clinical studies have been performed to date.
The authors of the CRASH-2 study reported no difference in the rate of vascular occlusive events between groups [PE 72 (0.7%) for the TXA group vs. 71 (0.7%) for the control group; DVT 40 (0.4%) for the TXA group vs. 41 (0.4%) for the control group] or risk of stroke [57 (0.6%) for the TXA group vs. 66 (0.5%) for the control group], but in this report the number of these events was small, and no standard diagnostic method was presented. The MATTERs study, where TXA was used in the military, documented no difference in thromboembolic events between groups [43]. In the STAAMP trial [33], an RCT on the effects of prehospital use of TXA in injured patients with hemorrhagic shock, the incidence of deep vein thrombosis, and pulmonary embolism was 2.7:1.5% (p = 0.83) and 2.9%:1.5% (p = 0.78) in the TXA and placebo groups, respectively, and there was no significant difference between the 2 groups.
In the international, multicenter, randomized trial on the effect of TXA on the thromboembolic events in patients with gastrointestinal bleeding (HALT-IT), the incidence of arterial thromboembolic events (myocardial infarction or stroke) in the TXA treated group was similar to the control group, and the venous thromboembolic events (DVT or pulmonary embolism) occurred significantly more frequently than in the control group [42]. However, it should be taken into consideration that the TXA dose in this study was slightly higher than the 24-hour dose compared with the CRASH-2 trial, and this result helped to decide the safe dosage of TXA.
A recent systematic review involving 216 trials and a total of 12,550 people, determined that there was no association between TXA and risk for total thromboembolism [risk difference = 0.001 (95%); CI −0.001–0.002; p = 0.49] including deep vein thrombosis, pulmonary embolism, myocardial infarction or ischemia, and cerebral infarction or ischemia [63].
However, some studies have shown that the administration of TXA in trauma patients is an independent risk factor for the venous thromboembolism [64–67].
In addition, a recently published meta-analysis review including 234 studies, reported seizures increased in patients receiving more than 2 g/day of TXA [3.05 (1.01–9.20)] [68]. Meta-regression showed an increased risk of seizures with increased dose of TXA (p = 0.011) indicating a high dose of TXA use should be avoided because there may be dose-dependent increase in the risk of seizures [68].
10. Value and limitations of the CRASH-2 trial
The CRASH-2 trial [3] was a large scale, international, multicenter randomized placebo-controlled trial on the effect of TXA use on mortality, vascular occlusive event, and transfusion in the adult trauma patients. This trial enrolled 20,211 injured adults with significant bleeding or shock. All-cause 28-day mortality was reportedly 1,463 (14.5%) in patients who received TXA, and 1,613 (16.0%) in patients who received the placebo [RR 0.91 (95% CI 0.85–0.97; p = 0.0035)]. There was no difference in the rate of vascular occlusive events between groups. In this subgroup analysis for the effectiveness of the TXA, the most beneficial subgroup was use within 3 hours from injury in the patients with the most severe shock (SBP ≤ 75 mmHg). This result of subgroup analysis has provided many implications regarding the effectiveness of TXA use in trauma patients.
The relationship between TXA administration time and mortality benefit in trauma patients can be interpreted by considering fibrinolysis phenotype. Considering the basic pharmacological action of TXA, an antifibrinolytic agent, which shows an effective beneficial effect on patients with hyperfibrinolytic status, and the results of several studies showing that the hyperfibrinolytic phenotype is more common in cases of severe damage or shock, or in the early stages of injury [61,69], the result of subgroup analysis of CRASH-2, regarding the timing of TXA administration, are fully appropriate and have implications. However, despite the insight that the CRASH-2 trial gives us, it should be interpreted with several caveats in mind. That is, there is no consideration of the international differences in trauma systems, prehospital infrastructure, and resources between participating countries, and no data regarding the effect of pre-hospital interventions, no formal stratification of severity of injury, and not all patients were severely injured. In addition, the CRASH studies lack assessment of fibrinolysis or coagulation.
11. Goal directed treatment of TIC and use of TXA
TIC appears in various clinical manifestations after injury, ranging from hypocoagulability to hypercoagulability, and this is explained by a complex mechanism involving the cell-based hemostasis concept including platelet function, endothelial cell dysfunction, and dysregulation of fibrinolysis. Clinically, a quick and accurate evaluation of the function and amount of coagulation factors, and the fibrinolysis status of a patient with coagulopathy is crucial in selecting the appropriate blood component, and deciding whether to administer TXA.
However, the plasma-based conventional coagulation test has many weaknesses in determining these various blood coagulation and fibrinolysis conditions. On the contrary, the viscoelastic hemostatic assay (VHA), like thromboelastography (TEG) or rotational thromboelastography (ROTEM), can provide better information about the coagulability status as a result of dysfunction of the platelet or deletion of some coagulation factors. In particular, it is superior in determining the state of fibrinolysis [70]. In a study that compared 2 diagnostic methods, TEG data was clinically superior to the results from 5 conventional coagulation tests. In addition, TEG identified patients early that had an increased risk of requiring RBC, plasma and platelet transfusions, and fibrinolysis, and it was reported that TEG can replace the conventional coagulation tests [71]. Many researchers have explored the threshold values of several parameters of VHA, which has better sensitivity and specificity for TIC diagnosis compared with the conventional tests [72].
In addition, to VHA, research on the fibrinolysis phenotype of patients with TIC is emerging that supports individualized goal-directed resuscitation after injury [73,74].
Gonzalez et al [75] reported that resuscitation of severe trauma patients using a goal-directed, TEG-guided massive transfusion protocol resulted in significant survival benefit compared with the preemptive strategy using conventional coagulation assays (CCA) 19.6%, 36.4%, respectively (p = 0.049). Recently, VHA guided hemostatic resuscitation for severely injured patients was associated with better results than using CCA in terms of survival, massive transfusion, and volume of the transfusion [76].
“The European guideline on management of major bleeding and coagulopathy following trauma: 6th edition” revised in 2019 recommended resuscitation measures using goal-directed strategy guided by standard laboratory coagulation values and/ or viscoelastic measures (VEM) [30].
While research on the superiority of goal-directed resuscitation based on VHA has primarily focused on survival rates and transfusion volumes, there is a notable lack of information guiding the use of TXA. This highlights a critical gap in our understanding that warrants further investigation [77].
The state of fibrinolysis can be determined by measuring LY30 in TEG (the percentage reduction in the area under the curve at 30 minutes after maximal amplitude) and Li30 in ROTEM [the residual clot firmness at 30 minutes after clotting time (CT)] [7]. It appears that a certain degree of consensus has been reached regarding the range of measurement values for each fibrinolysis phenotype. That is that HF is LY30 ≥ 3% and Li30 is > 15%, physiological fibrinolysis is LY30 0.9–3% and Li30 5–15%, and fibrinolysis SD is LY30 < 0.9% and Li30 < 5% [5,7].
These measurements theoretically inform us about the most appropriate indication of TXA for the coagulopathy. Moore et al [7] advocated that TXA should only be used if patients have VHA evidence of hyperfibrinolysis. In the prehospital setting, TXA use should be used prudently in severely injured patients with shock, based on the belief that shock is the main driver of fibrinolytic dysregulation.
Conclusion
TXA has emerged as a crucial hemostatic agent in the management of trauma patients, demonstrating a reduction in mortality and bleeding, particularly when administered early, and in patients with shock or requiring massive transfusion. However, the effects of TXA can vary depending on the severity of the injury and its location, such as in cases of brain injury.
The use of VHA for assessing the fibrinolysis status of TIC has been highlighted as a valuable tool for guiding the appropriate use of TXA. This approach allows for individualized treatment strategies that can optimize patient outcomes.
However, it is important to note that in cases of severe hemorrhagic shock, the patient’s physiology and clinical presentation may be more critical than waiting for the results from VHA or conventional coagulation assays in guiding initial resuscitation strategies, including the administration of TXA.
While significant strides have been made in understanding the role and application of TXA in trauma management, further research is needed to fully elucidate its optimal dosage, timing, safety, and efficacy across different types of injuries. This underscores the need for ongoing investigation into this therapeutic agent within the context of comprehensive, patient-centered trauma care.
Acknowledgment
I acknowledge the significant contributions of Hyerim Lee, RN and Jiwon Kim, RN to this review article. Their diligent efforts in searching, collecting, sorting, and referencing the literature and data have been invaluable. Their dedication and hard work have greatly enhanced the quality of this work. I extend my deepest gratitude for their assistance.
Notes
Conflicts of Interest
The author has no conflicts of interest to declare.
Funding
None.
Ethical Statement
This research did not involve any human or animal experiments.
Data Availability
All relevant data are included in this manuscript.